NanoVation announces new NHP data
Long-circulating LNP (lcLNP™) unlocks ligand-free in vivo CAR
NanoVation announces new NHP data
Long-circulating LNP (lcLNP™) unlocks ligand-free in vivo CAR
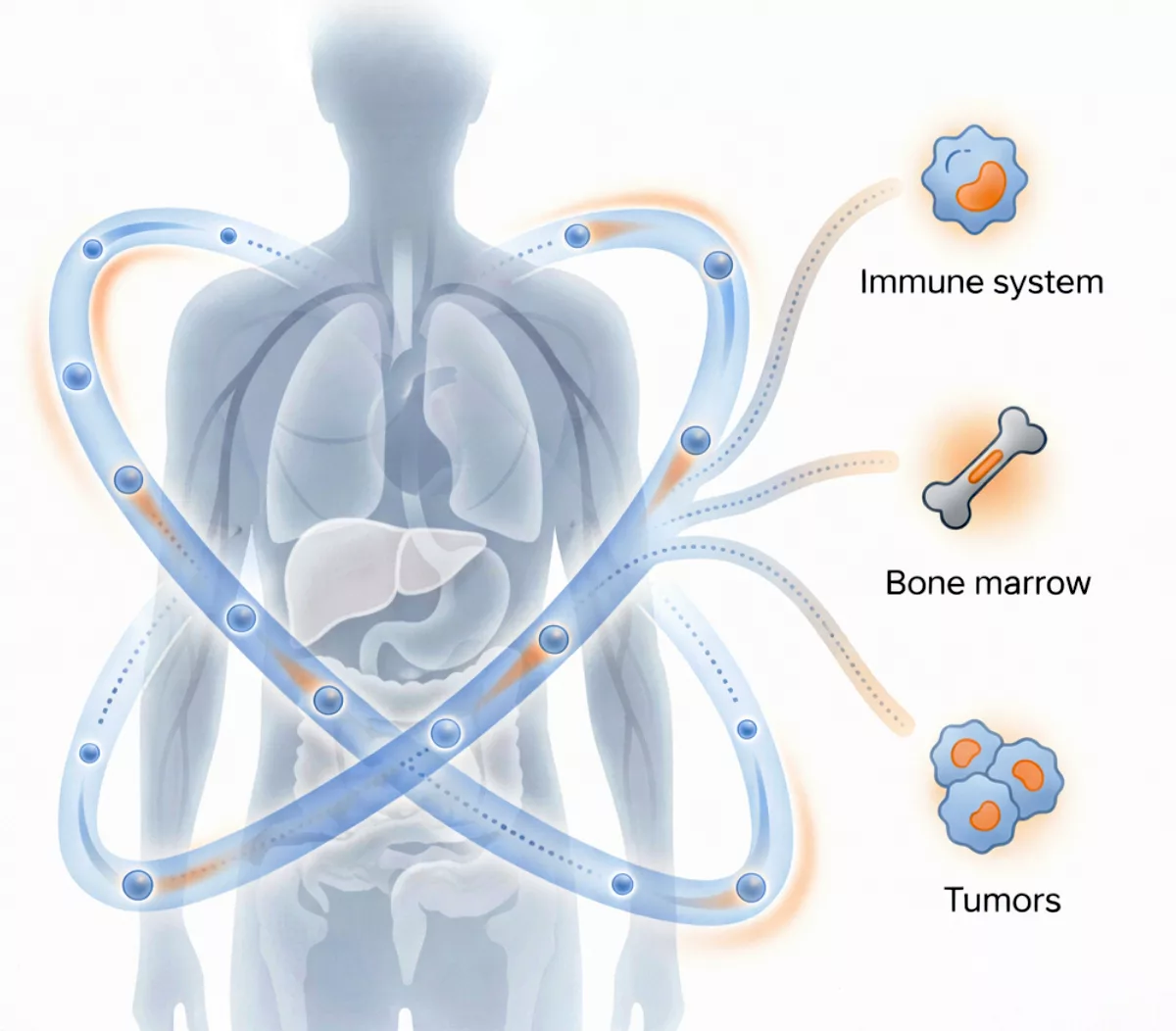
Delivery beyond the liver
New mouse & NHP data
further highlight NanoVation’s ligand-free, lcLNP™ technology for functional delivery to extrahepatic tissues. Demonstrating in vivo CAR for B cell depletion at clinically relevant doses.
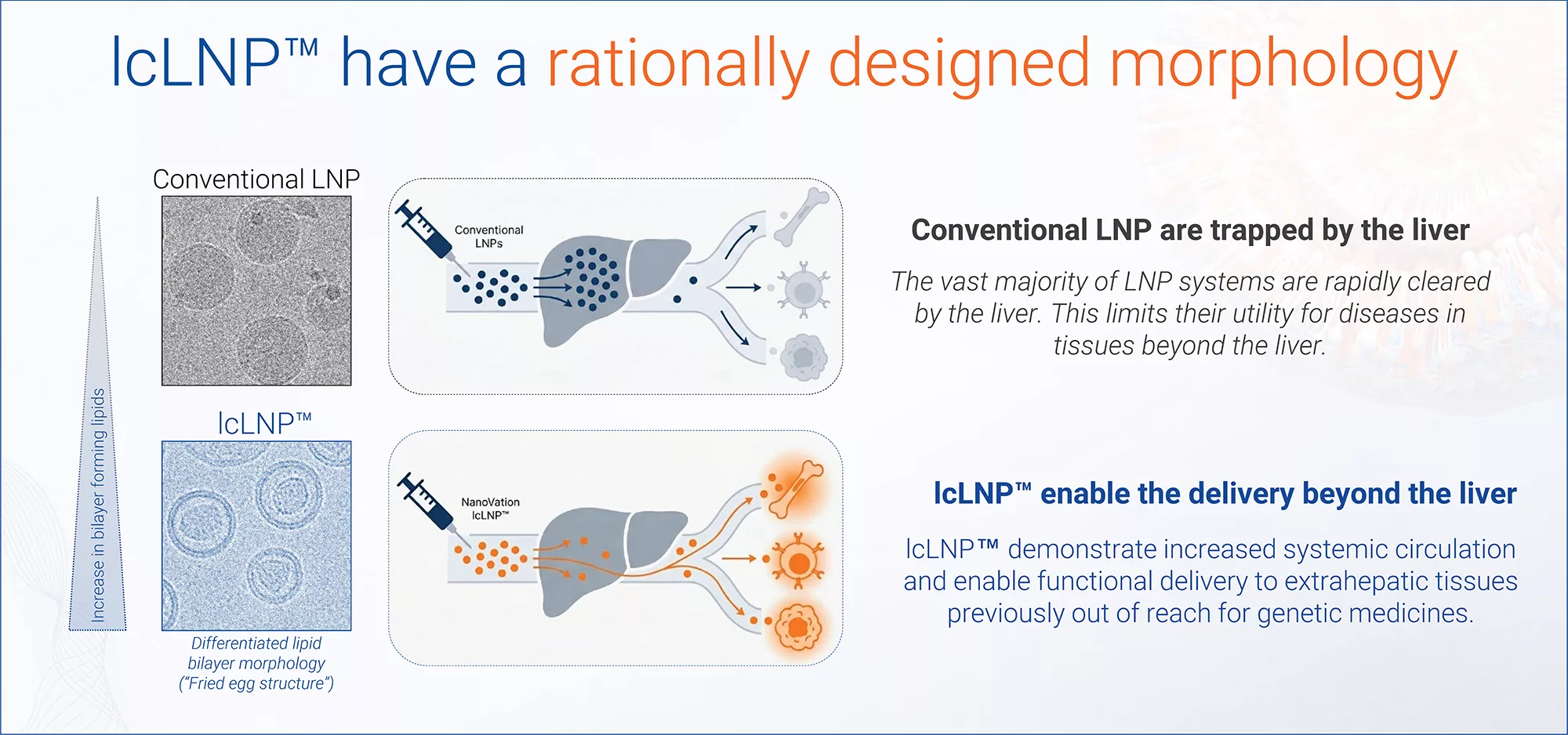
Conventional LNPs are quickly cleared by the liver…
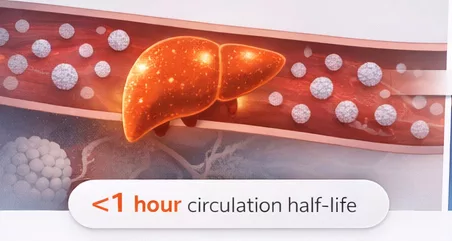
…NanoVation’s lcLNP™ circulate longer, enabling delivery beyond the liver

Delivery beyond the liver starts with longer circulation
Most LNPs are cleared too quickly to reach extrahepatic targets.
NanoVation’s lcLNP™ is engineered to stay in circulation longer, enabling functional delivery to immune compartments, bone marrow, tumors and beyond.
Engineered LNP morphology
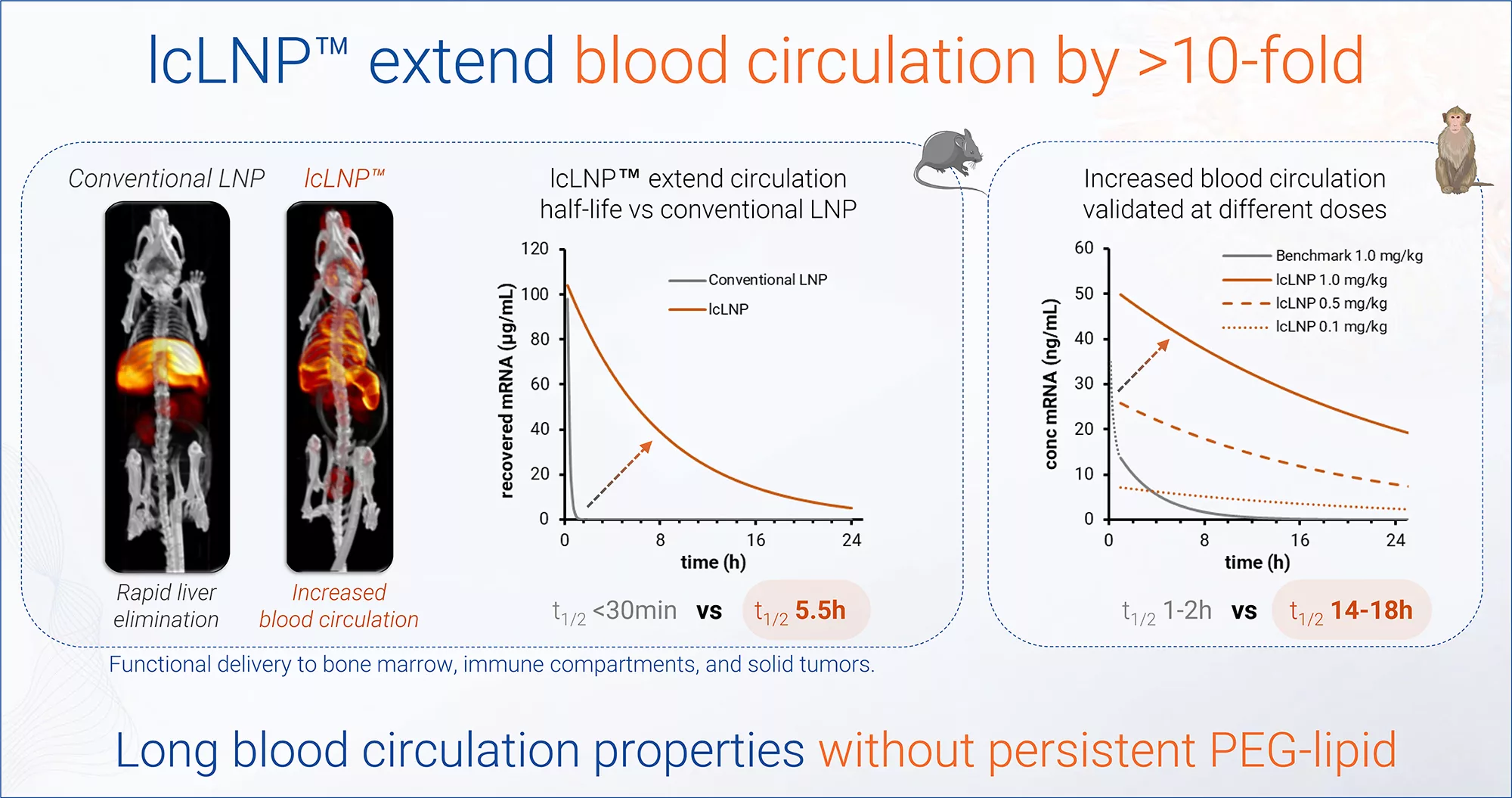
Extended blood circulation without persistent PEG-lipids overcoming rapid hepatic clearance
Extended systemic exposure
Functional delivery to extrahepatic tissues previously out of reach for genetic medicines
Improved therapeutic index
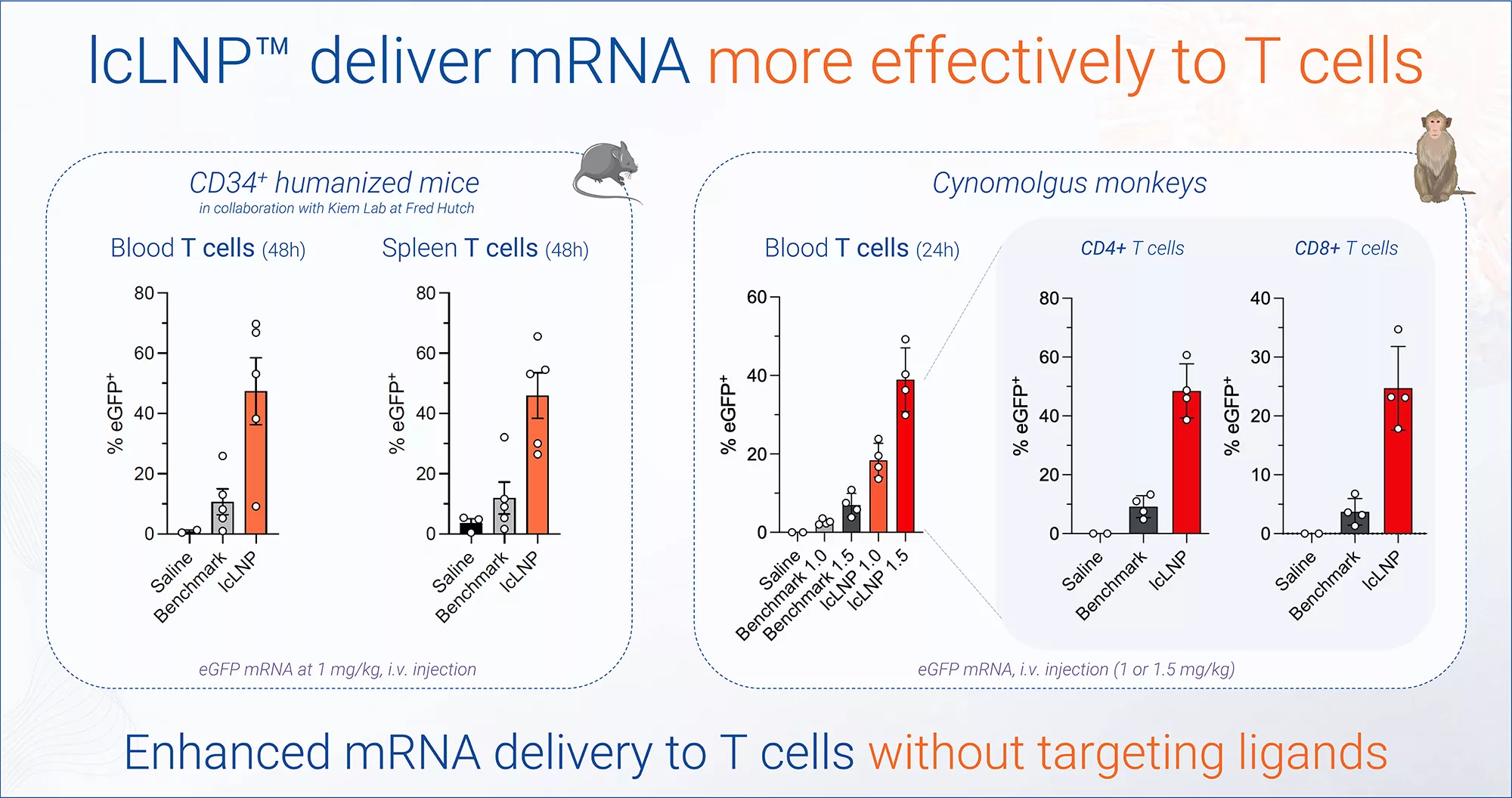
Efficient and safe delivery to specific cell types without targeting ligands at clinically relevant doses

Engineered LNP morphology
Extended blood circulation without persistent PEG-lipids overcoming rapid hepatic clearance

Extended systemic exposure
Functional delivery to extrahepatic tissues previously out of reach for genetic medicines

Improved therapeutic index
Efficient and safe delivery to specific cell types without targeting ligands at clinically relevant doses

Engineered LNP morphology
Extended blood circulation without persistent PEG-lipids overcoming rapid hepatic clearance

Extended systemic exposure
Functional delivery to extrahepatic tissues previously out of reach for genetic medicines

Improved therapeutic index
Efficient and safe delivery to specific cell types without targeting ligands at clinically relevant doses
Key results lcLNP™ enabling in vivo CAR-T
Contact our business development team

Business Development

Business Development
Preclinical data
Enabling in vivo CAR
for complete B cell depletion at clinically relevant dose (0.1 mg/kg)
Preclinical data
Improved safety profile
demonstrated in mice and NHP
no indication of liver or kidney toxicity
no changes in hematological factors
no changes in cytokines
allowing higher & repeat dosing

Extended blood circulation
without persistent PEG-lipids
Up to 18h circulation half-life in NHP (vs <1h for conventional LNPs)
This extended exposure underpins functional delivery to immune compartments and bone marrow, including enhanced mRNA delivery to T cells – extrahepatic targets that partners have struggled to reach without targeting ligands.