NanoVation’s ligand-free lcLNP™ achieves complete B cell depletion in NHP at 0.1 mg/kg
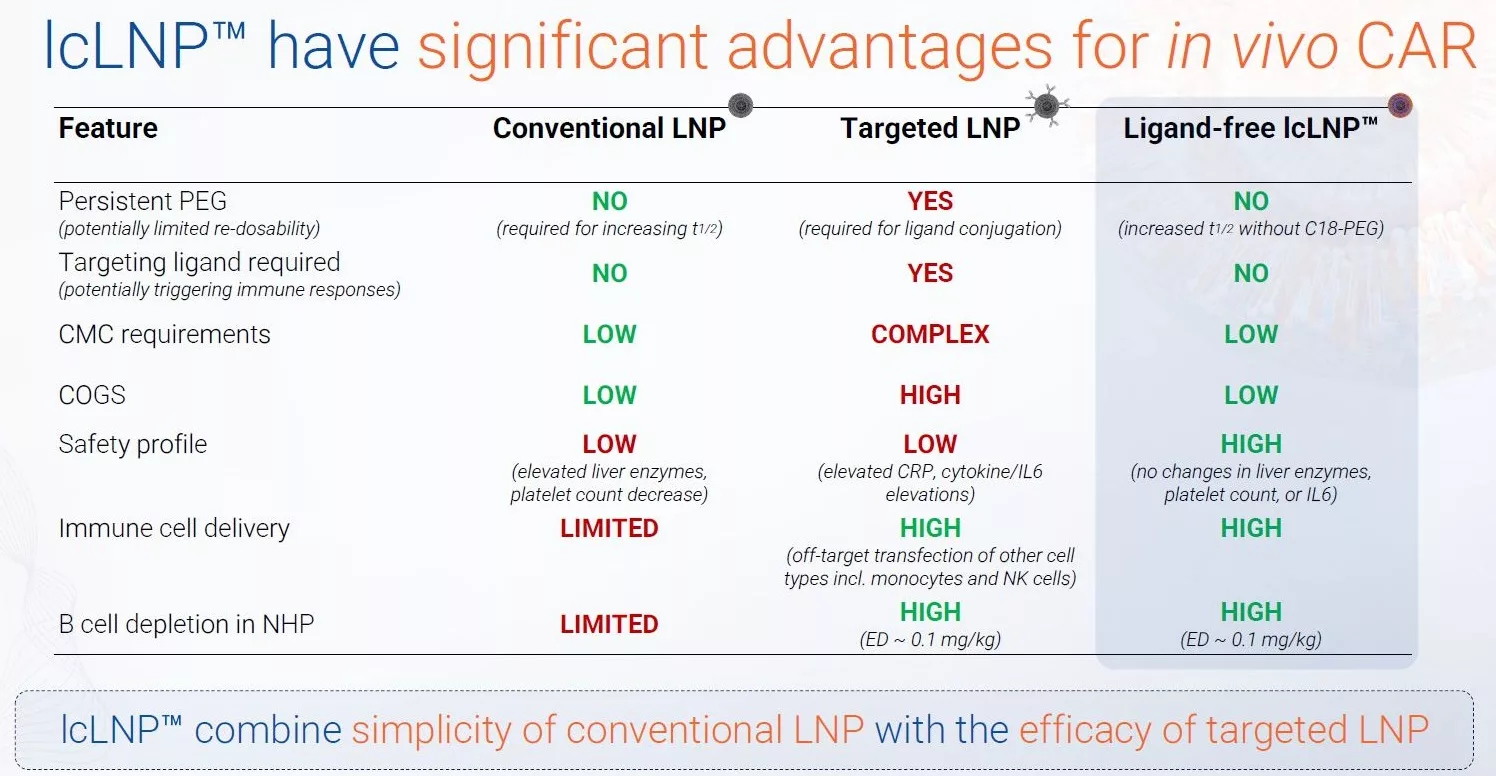
Targeted LNPs can reach new tissues, but they require exogenous ligands – introducing immunogenicity considerations as well as manufacturing and regulatory complexities that slow programs down.
Why does this matter?
Extrahepatic delivery remains one of the most significant bottlenecks for mRNA therapeutics. While the success of mRNA COVID-19 vaccines and Onpattro (as the first-ever siRNA therapeutic) demonstrated the power of LNP delivery, those LNPs do not allow for delivery beyond the liver – a natural endpoint for conventional LNPs. For therapeutic areas like autoimmune disease, oncology, and in vivo cell therapy, the payload needs to reach immune cells, bone marrow, or tumors.
Until now, achieving this has required persistent PEGylation to increase systemic circulation as well as complex targeting strategies involving ligand conjugation, which introduces manufacturing challenges, immunogenicity risks, and regulatory hurdles that can slow clinical development.
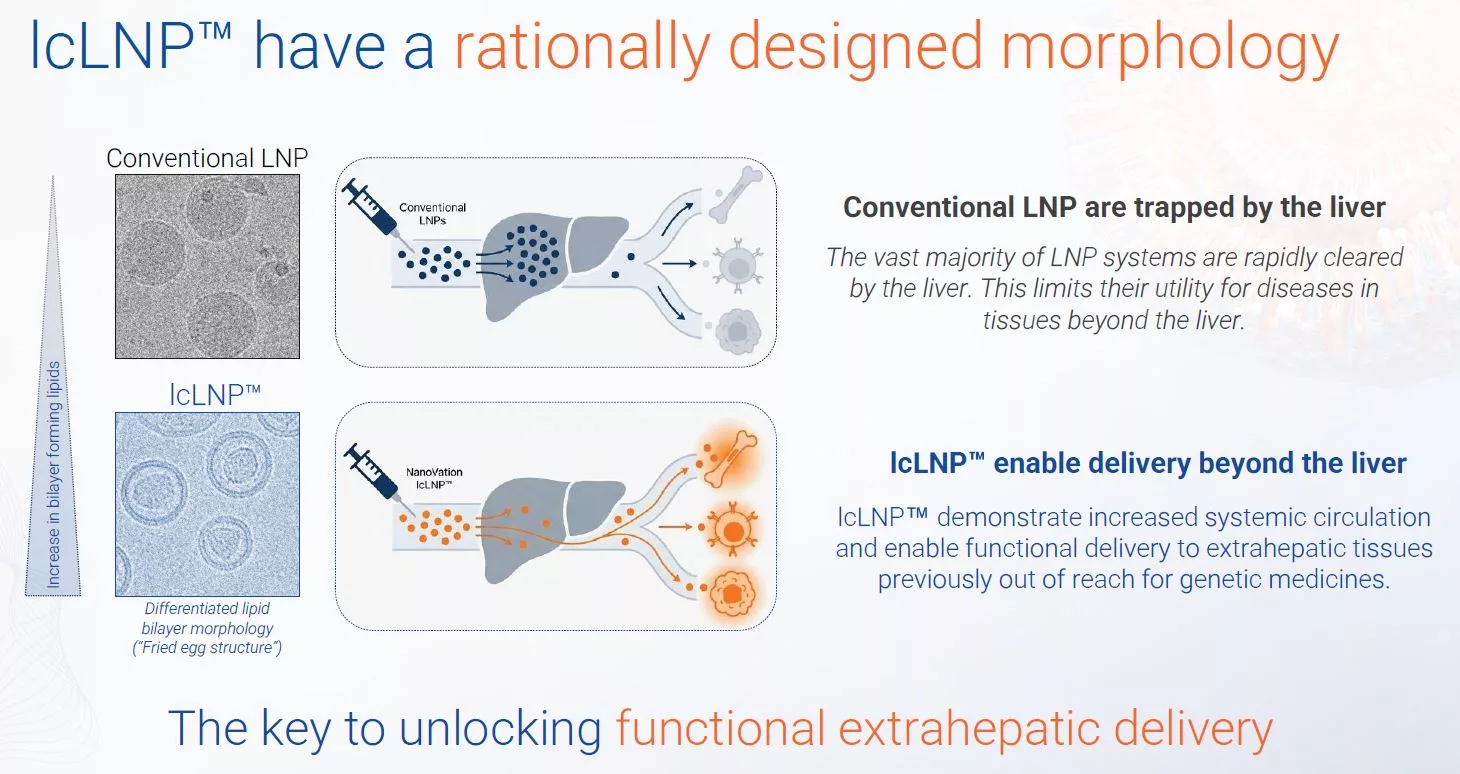
NanoVation’s ligand-free lcLNP™ (long-circulating LNP) technology extends where genetic medicines can go, with demonstrated extrahepatic delivery, while avoiding the pitfalls of added ligands.
Instead of adding targeting ligands, lcLNP™ morphology is engineered to extend systemic exposure and become long-circulating, with circulation half-lives of up to 18 hours in non-human primates (NHP), compared to less than one hour for conventional LNPs.
This extended exposure underpins functional delivery to bone marrow and immune compartments, including efficient mRNA delivery to T cells – extrahepatic targets drug developers have struggled to reach.
New mouse and NHP data show that the lcLNP™ platform enables ligand-free in vivo CAR.
In a preclinical lupus mouse model, lcLNP™-based anti-CD19 CAR delays disease onset and improves survival at low doses. In NHP, lcLNP™-based anti-CD20 CAR drive clear, concentration-dependent B cell depletion with an effective dose of 0.1 mg/kg for complete B cell depletion in peripheral blood – on par with targeted LNP systems.
Across studies, lcLNP™ have demonstrated a favourable tolerability profile, including stable liver and kidney markers and >50-fold lower IL-6 cytokine levels than comparator targeted LNPs – all achieved without persistent PEG lipids or targeting ligands, reducing the immunogenicity and redosing risks often associated with targeted LNP platforms.
For our partners, our lcLNP™ platform offers a practical path to extrahepatic genetic medicines – including in vivo CAR – that can combine targeted-like efficacy with the simplicity, manufacturability and scalability of conventional LNPs.
NanoVation has presented this new data at RNA Leaders Europe 2026. More data will be presented at the American Society of Gene & Cell Therapy (ASGCT) 2026 Annual Meeting.
About NanoVation Therapeutics
NanoVation Therapeutics is a Vancouver-based biotechnology company developing long-circulating lipid nanoparticle (lcLNP™) technology for genetic medicines. Founded on proprietary lipid chemistry and LNP engineering expertise, NanoVation’s platform enables functional delivery to extrahepatic tissues including immune cells, bone marrow, and tumors, without the need for targeting ligands. The company partners with pharmaceutical and biotech organizations to advance xRNA, DNA, and gene editing programs that require delivery beyond the liver.